Abstract
Loss of function mutations in TET2 are commonly found in clonal hematopoiesis and myeloid malignancies. Cells with TET2 loss have demonstrated heightened responses to inflammatory ligands, such as LPS and TNFα, and TET2KO mice exhibit a myeloproliferative phenotype and increased stem cell self-renewal.
Here, we investigated this dysregulated signaling with conditionally immortalized Hoxb8-ER TET2KO progenitors, an in vitro model that allows differentiation of progenitors upon removal of estrogen from the media. We used ATAC-Me, a method which provides chromatin accessibility (ChrAcc) and DNA methylation (DNAme) data from the same DNA fragment library, along with phosphoflow cytometry, RNAseq, and qRT-PCR to investigate the effects of TNFα or IL-1β stimulation on TET2KO cells. We also tested the effect of IL-1β exposure on the expansion of primary WT and TET2KO HSCs.
First, we used ATAC-Me to study the effects of TET2 loss on myeloid differentiation. In differentiated cells, we found that 1204 regions gained ChrAcc in both genotypes. Out of these, 149 regions were significantly hypermethylated in TET2KO cells(p=1.329e-5), meaning they do not become demethylated in TET2KO cells. Gene ontology (GO) analysis of these regions revealed that they were significantly enriched for the terms "abnormal hematopoietic system physiology, "abnormal blood cell physiology", and "abnormal adaptive immunity", including inflammation linked genes like Csf1, Csf2rb, Tlr2, and Tlr6.
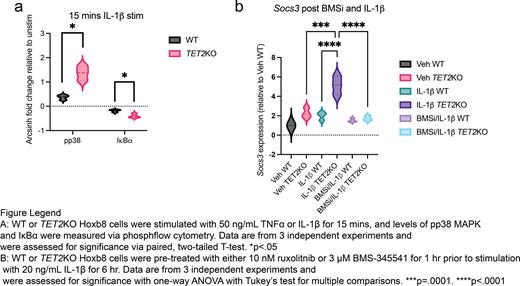
This led us to investigate differences in cytokine signaling responses with phosphoflow cytometry. We stimulated WT or TET2KO Hoxb8 cells with TNFα or IL-1β for 15 mins and saw statistically significant differences in levels of pp38 MAPK and IκBα with IL-1β, but not TNFα (Fig. 1A, p=.01 for pp38, p=.04 for IκBα). We then sought to determine the TNFα or IL-1β driven transcriptional programs in TET2KO cells with bulk RNAseq after 6 hours of stimulation.
Visualization of principal component analysis of RNAseq data demonstrated that IL-1β stimulation drove large global transcriptome changes in TET2KO cells versus WT, compared to smaller differences between TET2KO and WT in unstimulated and TNFα treated cells. IL-1β stimulation in TET2KO vs WT cells also caused significant enrichment in several genesets: HALLMARK_TNFA_SIGNALING_VIA_NFKB (Progenitor FDR=.02,Differentiated FDR=.02) HALLMARK_IL6_JAK_STAT3_SIGNALING (Progenitor FDR=.04), and HALLMARK_INFLAMMATORY_RESPONSE (Progenitor FDR=.03, Differentiated FDR=.04) with immune inhibitors, including Socs3, Nfkbia, Tnfaip3, and inflammatory cytokines including Il6, Tnf, Il1b, and Ccl2,driving the enrichment, many of which we confirmed via qRT-PCR.
Given its role in resolving inflammation and increase in expression in TET2KO cells, we tested the kinetics of Socs3 expression after IL-1β stimulation. During a 24-hour time course after IL-1β stimulation, Socs3 expression was significantly elevated in TET2KO compared to WT as early as 1 hour and remained elevated through 24 hours. During this same time course, we found that Csf2, the gene that encodes granulocyte-monocyte colony stimulating factor (GM-CSF) was massively increased in the TET2KO cells over WT cells. Thus, we identified elevated genes that modulate JAK-STAT signaling (Socs3 and Csf2) downstream of IL-1β signaling in TET2KO cells. We also tested the expansion of sorted HSCs from WT or TET2KO mice with IL-1β and saw that IL-1β caused expansion in TET2KO but not WT cells. To confirm that Socs3 was directly downstream of IL-1β-IκBα-NFκB, and not a secondary signal through JAK-STAT, we treated WT or TET2KO cells with ruxolitinib or BMS-345541 (JAK1/2 and IκBα kinase (IKK) inhibitors, respectively) before IL-1β stimulation. We found that levels of Socs3 were significantly lower in TET2KO cells with IKK inhibition, but not JAK inhibition, pointing to a direct upregulation of Socs3 in response to IL-1β (Fig. 1B, p<.0001).
Our data demonstrate that the epigenetic effects of TET2 loss heighten response to inflammatory signals, specifically IL-1β. This manifests as increased phosphorylation of downstream signaling proteins and differential transcriptional responses to IL-1β. Inhibition of IKK-mediated IκBα phosphorylation, but not JAK1/2 inhibition, results in almost complete reduction of Socs3 transcription. Future studies in our lab will be done to assess the role of Socs3 on the fitness of TET2KO cells.
Disclosures
Ferrell:Incyte: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal